 
Polysaccharides are classified into two parts, namely A molecule of a polysaccharide has n number of sugar molecules bound together to form a larger molecule. 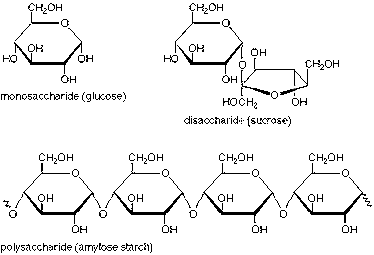
In the human body, several enzymes known collectively as amylases degrade starch sequentially into usable glucose units.Ĭellulose, a fibrous carbohydrate found in all plants, is the structural component of plant cell walls.These are complex carbohydrates made up of repeating units of monosaccharides that are attached together by Glucosidic linkage. The complete hydrolysis of starch yields, in successive stages, glucose: Dextrins are more easily digested than starch and are therefore used extensively in the commercial preparation of infant foods. Because of their characteristic stickiness with wetting, dextrins are used as adhesives on stamps, envelopes, and labels as binders to hold pills and tablets together and as pastes. The shine and stiffness imparted to clothing by starch are due to the presence of dextrins formed when clothing is ironed. These branch points occur more often in glycogen.ĭextrins are glucose polysaccharides of intermediate size. 
Both amylopectin and glycogen contain branch points that are linked through α-1,6-linkages. The helical structure of amylopectin is disrupted by the branching of the chain, so instead of the deep blue-violet color amylose gives with iodine, amylopectin produces a less intense reddish brown.įigure 5.1.2: Representation of the Branching in Amylopectin and Glycogen. A molecule of amylopectin may contain many thousands of glucose units with branch points occurring about every 25–30 units (Figure 5.1.2). (b) Because of hydrogen bonding, amylose acquires a spiral structure that contains six glucose units per turn.Īmylopectin is a branched-chain polysaccharide composed of glucose units linked primarily by α-1,4-glycosidic bonds but with occasional α-1,6-glycosidic bonds, which are responsible for the branching. (a) Amylose is a linear chain of α-D-glucose units joined together by α-1,4-glycosidic bonds. This color test is sensitive enough to detect even minute amounts of starch in solution.įigure 5.1.1: Amylose. The characteristic blue-violet color that appears when starch is treated with iodine is due to the formation of the amylose-iodine complex. When coiled in this fashion, amylose has just enough room in its core to accommodate an iodine molecule. Experimental evidence indicates that amylose is not a straight chain of glucose units but instead is coiled like a spring, with six glucose monomers per turn (part (b) of Figure 5.1.1). Amylose is a linear polysaccharide composed entirely of D-glucose units joined by the α-1,4-glycosidic linkages we saw in maltose (part (a) of Figure 5.1.1). Natural starches consist of about 10%–30% amylase and 70%–90% amylopectin. 
Starch is a mixture of two polymers: amylose and amylopectin. We often think of potatoes as a “starchy” food, yet other plants contain a much greater percentage of starch (potatoes 15%, wheat 55%, corn 65%, and rice 75%). The breakdown of starch to glucose nourishes the plant during periods of reduced photosynthetic activity. It occurs in plants in the form of granules, and these are particularly abundant in seeds (especially the cereal grains) and tubers, where they serve as a storage form of carbohydrates. Starch is the most important source of carbohydrates in the human diet and accounts for more than 50% of our carbohydrate intake. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
